It is often said that the 21st century will be (is) the age of biology; much like the previous century was for physics. New discoveries are occurring and biological information is growing both in size and complexity at an exponential rate. A major factor fueling this growth is the plethora of technologies available to the modern biologists in their quest to uncover the very basic molecular mechanisms of life.
 green fluorescent protein (GFP).
green fluorescent protein (GFP).

The jellyfish, Aequorea victoria, on the left and green bioluminescence observed around the margin (note the picture on the left does not show fluorescence !)
However, it took another thirty years before the GFP became the almost ubiquitous cellular and molecular biology tool it is today. In 1987, Doug Prasher, then at the Woods Hole Oceanographic Institute, discovered and was able to make a copy of the DNA sequence within the jellyfish gene that encoded for GFP. He did not, however, succeed in making a glowing protein from the DNA sequence in the lab. Subsequently, Prasher sent his sequence to a researcher at  typical trick used by biologists to make proteins. As shown in the figure on the right from Chalfie's work, the bacteria containing the genes for GFP (on the right side of the plate) emits green light under illumination with ultaviolet lamp (it was a graduate student doing rotation in Chalfie’s lab that actually performed the work and made the discovery!). This seminal work, published in 1994 in the journal Science, led to 'an explosion of color' in the biological world. Subsequently, other researchers showed that the GFP could be produced, alone or in tandem with other proteins in a variety of organisms.
typical trick used by biologists to make proteins. As shown in the figure on the right from Chalfie's work, the bacteria containing the genes for GFP (on the right side of the plate) emits green light under illumination with ultaviolet lamp (it was a graduate student doing rotation in Chalfie’s lab that actually performed the work and made the discovery!). This seminal work, published in 1994 in the journal Science, led to 'an explosion of color' in the biological world. Subsequently, other researchers showed that the GFP could be produced, alone or in tandem with other proteins in a variety of organisms.
Over the last decade, a great deal of research has contributed towards understanding the underlying physical mechanisms of GFP’s light emission2 and importantly, towards improving its properties through genetic manipulation. The leader in this field has been Roger Tsien, who along with co-workers demonstrated that making small changes, such as replacing a few amino acids in GFP could make it glow brighter, mature faster and prevent aggregation of the protein inside cells. His group has also succeeded in tuning the absorption and emission of the original GFP through mutagenesis, leading to a veritable palette of fluorescent proteins that absorb and emit light through the entire span of the visible light spectrum (see below). Additionally, a Russian scientist, Sergey Lukyanov, used the GFP sequence as a 'bait' to search for novel fluorescent proteins in corals and succeeded in finding several GFP-like proteins, particularly a red-emitting fluorescent protein, dsRED from Anthozoa, which is also used widely.


Panel on top shows the fluorescent protein 'palette' developed by Tsien lab - note range of colors and the fruity names. On the left, artwork with bacteria expressing various colors of fluorescent protein.
The major advantage of GFP is that inside a living cell, it can emit light on its own without the help of another protein or other chemicals. It is also possible by using molecular biology techniques, to attach the DNA of GFP to the DNA of the protein of your choice to produce a recombinant DNA. When the information from such recombinant DNA gets translated into a protein within the cell, a tandem protein is created with the GFP unit hanging from the protein. Importantly, since the size of GFP is relatively small, in most cases it does not interfere with the regular functions of the protein it is attached to.
In the simplest of applications, after shining light on the cells, the total amount of fluorescence obtained from the cells provides a measure of the level of expression of the protein tagged with GFP. However the more useful applications involve cells placed under microscopes with high magnifying power (40x to 100x) in conjunction with either arc lamps or lasers for bright illumination and high-resolution detection devices such as CCD cameras for
bright illumination and high-resolution detection devices such as CCD cameras for
capturing images of the emitted light. In these cases, we can literally see where the protein of interest is located, or illuminate a particular subcellular structure. For example, the figure on the right shows the mesh of protein network that act as a 'skeleton' (in fact it is called the 'cytoskeleton') in majority of cells in higher organisms. A protein called 'actin' that is involved in this scaffold has been tagged with GFP.
It is also possible to tag two or more proteins in the cells with different fluorescent colors (see the fluorescent protein palette above) and follow their localization or movement in cells. This helps in noting where two proteins are localized  during a cellular function. In the figure to the right, the protein actin is now tagged with a cyan emitting fluorescent protein (CFP). Another protein, vinculin, has been tagged with a yellow fluorescent protein (YFP). You can observe that the YFPs are localized as small elliptical structures at many places. These are called focal adhesions, which form a link between the cell cytoskeleton (in cyan) and its extra-cellular matrix. This interaction help cells to adhere and eventually move about in a tissue.
during a cellular function. In the figure to the right, the protein actin is now tagged with a cyan emitting fluorescent protein (CFP). Another protein, vinculin, has been tagged with a yellow fluorescent protein (YFP). You can observe that the YFPs are localized as small elliptical structures at many places. These are called focal adhesions, which form a link between the cell cytoskeleton (in cyan) and its extra-cellular matrix. This interaction help cells to adhere and eventually move about in a tissue.
(in a future post, I will talk about a technique involving fluorescent proteins of two colors which is used to determine if two proteins interact with each other inside a cell)
Perhaps the most powerful application of fluorescent proteins is when you combine microscopy with time lapse video images. In such cases, it is possible to observe where and when the translocation of the protein in cells is taking place under a biological condition. For example, see a video here of a cell moving around with a protein involved in the focal adhesion tagged with GFP.
Visualization of protein location and dynamics in this manner enable scientists to place cells under various physiological conditions and observe the resultant phenotype of the protein behavior. Before the advent of GFP, scientist had to destroy the cells and use other tedious biochemical techniques to obtain similar information. Even then real-time data acquisition was not possible.
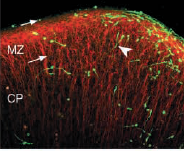
A quick search of the database will reveal more than ten of thousands of peer-reviewed publications where fluorescent proteins have been used to study protein functions at the cellular level. In most of these cases, research was conducted with either unicellular organisms or cells derived from tissues of mammals. However, apart from single cells, fluorescent proteins are also being used at the tissue and even the whole organism level. The picture below shows an example of a research which is investigating the movement of neurons (labeled with GFP) in the cerebral cortex.
A more well-known example of GFP in whole organisms, is the development of 'fluorescent mice' by the company Anticancer Inc . It is easy to follow tumor progression and cancer metastasis in such mice. Also, a Taiwanse reasearch group recently created 'fluorescent pigs'. Stem cells or organs from these pigs when transplanted into other organisms can be followed easily without requiring invasive techniques.


More examples of such applications of GFP can be found here. Apart from these animals, 'Alba' , the fluorescent rabbit and fluorescent aquarium fishes are two examples of more esoteric application of this scientific technology.
On a final note, betting markets for the Nobel Prize (yes they do exist !), were predicting this year’s Chemistry Nobel to go to Roger Tsein and others for their work on fluorescent proteins. It eventually went to Roger Kornberg for his work on DNA transcription. Considering the importance of fluorescence proteins and their wide-ranging revolutionary impact on biology, it is not far-fetched to think that the Nobel is not beyond the grasp of these researchers.
Notes:
1. Aequorin itself has been very useful for visualizing cellular calcium concentrations, the regulation of which is important for a number of physiological activities.
2. Without going into great details about physi-chemical mechanisms of GFP fluorescence, suffice to say that the protein has a barrel-like structure (see below); within the barrel, three critical amino acids are brought together in close spatial proximity, which forms the chromophore.
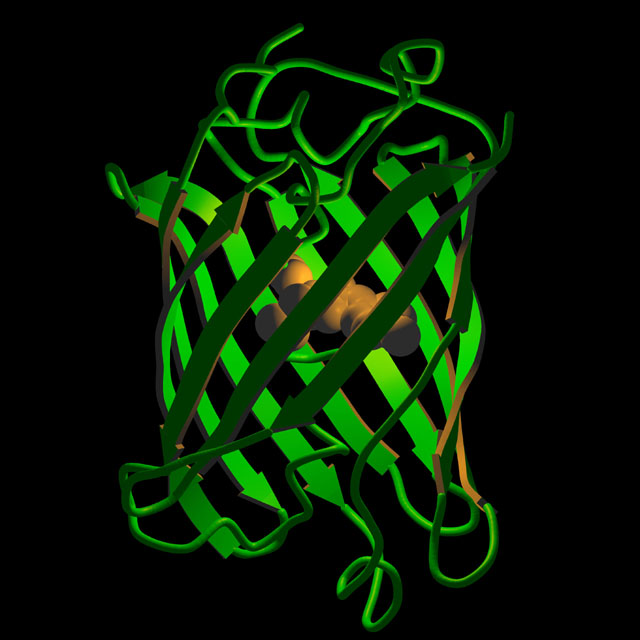
Artistic rendition of the three-dimensional structure of GFP.
4. Recommended further reading: This web-site is a very good resource for learning more about GFP's discovery, structure and applications. Also read this interview with Dr. Martin Chalfie.
Coming up: "Much to fret about ": on a technique known as fluorescence resonance energy transfer that enables biological distance measurements, detection of protein interactions, and can be used to look at protein functions at a single molecule level !
34 comments:
Awesome pictures! I loved the "bacteria art" :)
I was wondering... is the fluorescence on a DNA micro-array chip done using these proteins?
Prashanth: Yeah - those guys must have had fun while doing the bacterial art.
For DNA micro-arrays, organic fluorophores are used, not proteins.
Nice post on an increasingly important molecular biology tool... A couple of questions:
1. What is the function of GFP inside the cell when it occurs naturally?
2. What studies have been performed to show that in most cases, GFP does not affect the expression, cellular sublocalization, protein-protein interaction and functional efficiency of most proteins? I guess it is easier to prove functional efficiency but what about the rest because GFP is used as a probe in those studies too, right?
Libran:
1. GFP occurs naturally in the jellyfish in conjunction with Aequorin, the Ca sensing protein. As I mentioned, GFP absorbs the emitted blue light from Aequorin and emits green. What exactly is the functional relevance of this bioluminescence in jellyfish is not very well known, AFAIK.
2. As you mentioned, functionality can be tested easily e.g ion channels are often expressed as GFP fusion proteins - and in this case you simply measure currents through them in presence and absence of GFP.
Localization is more difficult to prove as such. With a lot of proteins, it has been confirmed with biochemical methods and I cannot recall any instance where GFP has mislocalized the protein. Also if you 'see' your protein in a localization not expected from predicted pathways, you obviously have to do further tests.
Eventually its a bit of the uncertainty principle - when you attach something to your protein and observe it - there is always some uncertainty about the tandem having exactly the same properties. But in many cases it is better to take that uncertainty over not being able to observe at all.
One known problem with GFP is its tendency (because of the small size) to get into the nucleus of the cell. This does not affect functions of the nucleus, and in majority cases where GFP is fused to a large protein it does not do this. But when fused to a small peptide - it is a nuisance for imaging - a problem I am grappling with now !
To address one of the issues that BaL brings up-
In budding yeast, GFP/YFP localization studies have been done with every known ORF. ANd in most cases, the localization is as predicted.
That is interesting... and thank you for answering my questions... Final question - is there any other technology being used currently for protein expression levels or for subcellular localization at the proteome level?
@ BaL - my model organism kicks ass..
Every protein in yeast has been tagged with a ProteinA -Calmodulin binding protein Tag and the expression level of each protein (using its endogenous promoter)has been quantified. And GFP/YFP studies show the localization.
Sakshi: Oh God ! NAYP (not another yeast person !).
Actually in my grad school we had two PIs who were really doing great work using yeast genetics. It became a running joke in the dept that if anyone ran into a difficult research problem or hit the wall, we would say 'oh - just use the AWESOME power of yeast genetics'.
No offense meant of course ! Yeasts are damn convenient buggers.
PS - don't they use Beta-galatosidase as another assay for expression levels (as well as in two-hydrid) ?
i LOVED the petri-plate art.
(yeah back-reading on a science blog can be fun too!! )
great post bongopondit. can't wait for you to FRET. :-)
What kind of lamp is used to exite the bacteria in the plate? Wavelenght? o_O
I really liked the text. And also, I agree with you, as the factor fueling, and this kind of things. So please keep posting articles like this one.
I'm so proud of the humanity and the different technological artifacts it has used to help with biological investigations. I have not idea which methods the scientists are using, but they are doing a very well job.
missouri car insurance You really make it seem so easy with your presentation but I find this topic to be really something which I think I would never understand. It seems too complicated and very broad for me. I am looking forward for your next post. Jason - texas auto insurance
What can I say about the blog .... It is only an issue to which we devote their time and attention to understand and learn more about this, actually I am in total agreement with what is written here and now I can only say good job guys continue doing its work..Actually have a very nice blog, I wish I could see everything you have all the time, I'm really entertained by your comments, and best wishes for your blog.
Thanks for discussing. Quite simple and straightforward to master. Done well!
Found your posting exactly when it wasreally needed. Thank you very much. It's already been very beneficial
Wonderful submit with many different wonderful written content! I guess you may be on the right way. Good luck
Amazing everyone!! This is just about the most fascinating write-up My spouse and i ever discovered! Very beneficial. Continue the good work.
Wonderful submit. Thank you for writing
Thanks for this post, I've been all the time searching for this convenient articels in all places but couldn't discover full information supplied here. thank you. and I'll just reference a few of you phrases .
Interesting article that you had posted... I couldn't ignore the new terms. but Optical engineering methodology uses optical methods to measure micro-vibrations with instruments like the laser speckle and others, it's all about new concepts and interferometer or to measure the properties of the various masse
Hey there! Would you mind if I share your blog with my twitter group?
There's a lot of people that I think would really enjoy your content. Please let me know. Thanks
My site - eco.kid
fsu russian dating scam http://loveepicentre.com/ dane cook and jessica alba dating
top [url=http://www.c-online-casino.co.uk/]uk casino bonus[/url] check the latest [url=http://www.casinolasvegass.com/]free casino[/url] unshackled no deposit hand-out at the foremost [url=http://www.baywatchcasino.com/]casino games
[/url].
Hmm is anyone else encountering problems with
the pictures on this blog loading? I'm trying to find out if its a problem on my end or if it's the blog.
Any suggestions would be greatly appreciated.
Review my web-site ; click through the following web site
good calories bad calories ebook http://audiobookscollection.co.uk/fr/Laung-Terng-Wang/m74844/ mighty words ebook publishing companies [url=http://audiobookscollection.co.uk/fr/A-Mind-Apart-Understanding-Children-with-Autism-and-Asperger-Syndrome/p227214/]drug guide ebook[/url] the rocket ebook
financial math ebook http://audiobooksworld.co.uk/Isaac-Markovich-Khalatnikov/m53068/ dark need free ebook [url=http://audiobooksworld.co.uk/fr/authors/?letter=Ha]free download sql notes documents ebook[/url] ebook blackstone
Hello! I'm at work surfing around your blog from my new apple iphone! Just wanted to say I love reading your blog and look forward to all your posts! Carry on the great work!
Also visit my webpage - www.rolexsale-shop.com
[url=http://certifiedpharmacy.co.uk/products/tricor.htm][img]http://onlinemedistore.com/5.jpg[/img][/url]
lake forest park city center pharmacy http://certifiedpharmacy.co.uk/products/atarax.htm pharmacy colleges in us [url=http://certifiedpharmacy.co.uk/products/tetracycline.htm]walgreens pharmacy brentwood ca[/url]
pharmacy technicians and math and help http://certifiedpharmacy.co.uk/categories/anti-acidity.htm walmart online pharmacy [url=http://certifiedpharmacy.co.uk/products/aricept.htm]aricept[/url]
appalachian school of pharmacy http://certifiedpharmacy.co.uk/products/tretinoin-cream-0-025-.htm walmart pharmacy orange city fl [url=http://certifiedpharmacy.co.uk/products/viagra-jelly.htm]target pharmacy mn[/url]
indiana ohio illinois online pharmacy http://certifiedpharmacy.co.uk/catalogue/z.htm rita wickham pharmacy venous access [url=http://certifiedpharmacy.co.uk/products/abana.htm]abana[/url]
orthodox online dating [url=http://freeinternetdating.info/matchmaker/free-matchmaker-service]free matchmaker service[/url] adult married dating ashely
dating sucks lousivile ky http://freeinternetdating.info/seeking/dancers-seeking-new-careers rainn wilson dating video
vin dicarlo dating diablo product review [url=http://freeinternetdating.info/matchmaker/frazier-the-matchmaker-clip]personal dating for animal lovers[/url] dating for single women online
I am no longer positive the place you're getting your info, but good topic. I needs to spend a while finding out much more or working out more. Thanks for wonderful information I used to be on the lookout for this info for my mission.
Feel free to visit my page :: voyeur sex
I like the helpful info you supply on your articles. I'll bookmark your weblog and check once more here frequently. I'm somewhat certain
I'll be told plenty of new stuff proper right here! Best of luck for the next!
Feel free to surf to my blog ... cam girls
After looking into a handful of the blog posts on your blog,
I seriously like your technique of writing a blog. I bookmarked it to
my bookmark webpage list and will be checking
back soon. Please visit my website as well and let me know your opinion.
Feel free to surf to my weblog ... http://www.cuteteenporn.net/video/29526/hottie-rides-big-stiff-dick.html
My brother recommended I would possibly like this website.
He was once totally right. This put up actually made my day.
You cann't believe just how so much time I had spent for this info! Thank you!
Here is my page; huis huren frankrijk
Post a Comment